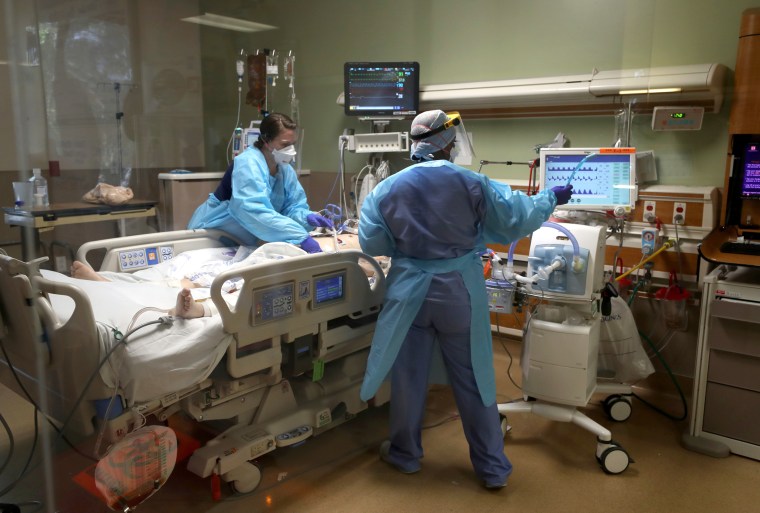
A common steroid drug that's been used for decades to treat conditions from altitude sickness to eye inflammation has been shown to reduce deaths by a third in the sickest patients in the hospital with COVID-19, British scientists say.
This is the first time, the researchers say, that a drug has been shown to have an effect on deaths rates of the virus that's killed more than 110,000 people in the U.S.
Full coverage of the coronavirus outbreak
Researchers at the University of Oxford in the U.K. compared outcomes of 2,104 hospitalized patients who received the steroid, called dexamethasone, with 4,321 patients who did not.
According to the researchers, deaths were reduced by about a third in those patients who were sick enough to require mechanical ventilation, and by about 20 percent among patients who had trouble breathing, but had not been put on a ventilator. Dexamethasone did not appear to help patients who did not require oxygen.
The results of the clinical trial, called Recovery, have not yet been published in a medical journal and the data have not been made available for outside experts to review. Researchers said they stopped the trial early because of the observed benefits.
Dr. Ashish Jha, director of the Harvard Global Health Institute, expressed frustration on Twitter that "it is now a feature of this pandemic that most findings made public via press release with little data to provide context." But, he added, "this is REALLY good news if it turns out to be true."
"The survival benefit is clear and large in those patients who are sick enough to require oxygen treatment," Dr. Peter Horby, one of the Recovery study researchers and a professor at the University of Oxford, said in a press release. "Dexamethasone is inexpensive, on the shelf, and can be used immediately to save lives worldwide."
Download the NBC News app for full coverage of the coronavirus outbreak
Doctors caution there is no evidence dexamethasone can prevent coronavirus infection, and should not be used as a prophylactic.
Outside experts called the results important and promising, but added the full data should be released for a complete analysis of how it worked in this clinical trial.
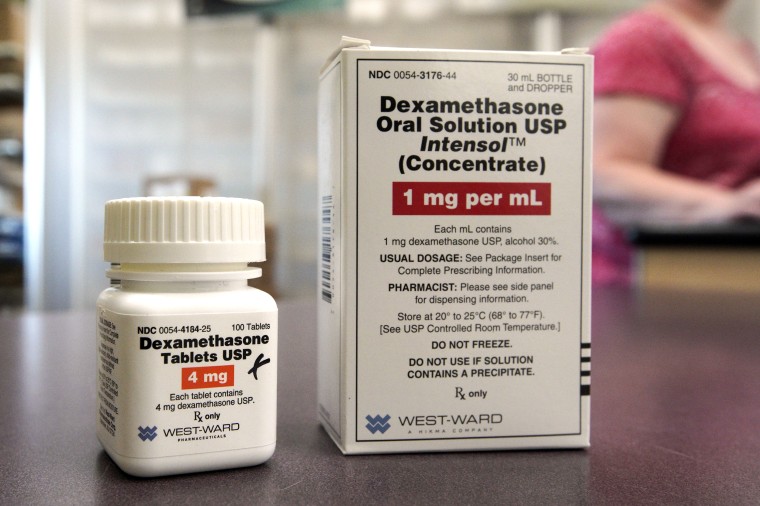
"The low cost and broad availability of this drug means that there is potential for considerable clinical impact by including it as part of standard treatment," Dr. Stephen Griffin, an associate professor of medicine at the U.K.'s University of Leeds, said in a statement. Griffin was not involved with the trial.
"Nevertheless, it will be important to assess how this important success might be further improved in order to increase patient survival, perhaps through the combination of low-dose dexamethasone with other inflammatory mediators, or with virus-targeted therapies, such as remdesivir," Griffin said.
Remdesivir is the only other therapy that's been shown in a clinical trial to help coronavirus patients. Results of that trial found that hospitalized patients who received the drug recovered more quickly and were able to be released from the hospital faster. It did not affect mortality, though.