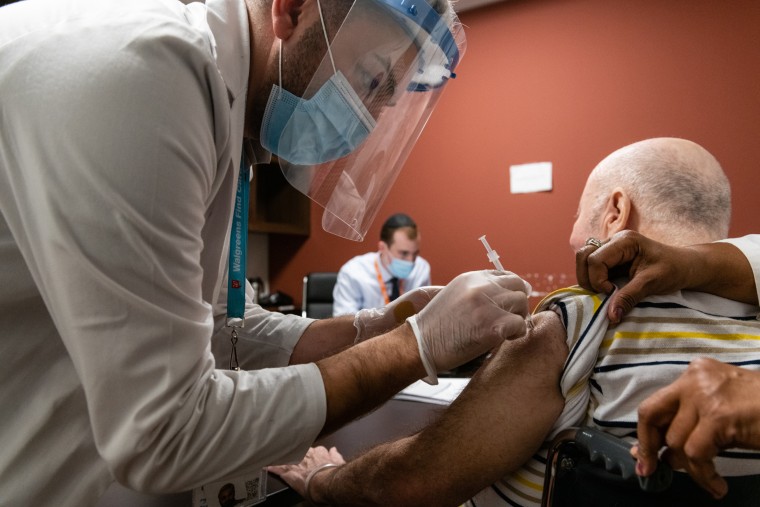
The Centers for Disease Control and Prevention reported Wednesday that at least 21 people have had a severe allergic reaction after receiving their first dose of the Covid-19 vaccine. All of the patients have recovered.
The reaction, called anaphylaxis, can be life-threatening, and must be treated immediately with an emergency injection of epinephrin, often given via EpiPen.
Full coverage of the coronavirus outbreak
The report included data from 1.9 million people who got the shot in the first week and a half it was available last month, from Dec. 14 to Dec. 23. That puts the rate of anaphylaxis at 11.1 cases per million doses given. For comparison, the rate of anaphylaxis following the flu shot is 1.3 per million, the CDC said.
"This is still a rare outcome," Dr. Nancy Messonnier, head of the CDC's National Center for Immunization and Respiratory Diseases, said during a media briefing Wednesday. "Right now, the known and potential benefits of the current Covid-19 vaccines outweigh the known and potential risks of getting Covid-19."
Still, the concern is great enough that the CDC is advising that those who had an immediate allergic reaction to the first dose of the vaccine not receive the second dose, Messonnier said.
The report comes the week many are scheduled to receive their second dose. During studies of the vaccine, side effects were more likely to be reported after the second shot.
The vaccine from Pfizer-BioNTech is given in two doses, separated by 21 days. According to company data, it takes those two shots for maximum effectiveness of about 95 percent. Moderna's vaccine is similarly effective, and given 28 days apart.
Since the report was finalized Dec. 23, eight more cases of vaccine-related anaphylaxis were confirmed, the CDC said, bringing the total number so far to 29. Most of those reactions were associated with the Pfizer vaccine, mainly because it rolled out a week ahead of the Moderna vaccine, so more doses have been given.
The CDC anticipates releasing additional information on reactions to the Moderna shots next week.
The Pfizer and Moderna vaccines are so far the only two vaccines available for use in the United States. As of Wednesday, 4.8 million doses had been administered.
Among the 21 cases for which details are available, the majority occurred quickly after the injection — within about 15 minutes. Women accounted for 90 percent of anaphylaxis cases. Many, but not all, had a history of allergies, and seven had previously suffered anaphylactic reactions.
Most required an emergency injection of epinephrine.
Download the NBC News app for full coverage of the coronavirus outbreak
Another 83 vaccine recipients had less severe allergic reactions, such as a rash, excessive itching and scratchy, irritated throats. Most of those occurred within a half hour of the shot. Similar to the patients who developed anaphylaxis, most of the individuals in these cases also had previously experienced other allergic reactions.
Messonnier said that researchers are working to find the cause of allergic reactions to either vaccine, but have not yet come up with anything definitive. There are no geographic similarities or links between any of the vaccine lots that would account for the reactions.
Overall, the CDC has received 4,393 reports of side effects or other reactions following the shot through the federal government's Vaccine Adverse Event Reporting System, or VAERS. Most were determined to be either unrelated to the vaccine or were not considered an allergic reaction. Vaccines are known to cause sore arms for a few days, even slight fever and fatigue. Those are not generally considered allergies.