A three-drug cocktail could be “life-changing” for patients with cystic fibrosis, researchers reported Thursday.
They found that adding a third drug to an existing combination of two drugs could improve the symptoms of cystic fibrosis patients, and might even help some of them live longer.
The big catch: the current drugs already cost around $300,000 a year and adding a third drug would likely boost that cost significantly.
Nonetheless, the findings are exciting for researchers who treat cystic fibrosis, a rare genetic disease that causes lung and digestive problems and shortens the lives of patients.
“These results represent a major breakthrough in cystic fibrosis therapeutics, with the potential for improving health and possibly survival in all patients who carry the most common CFTR mutation,” Dr. Fernando Holguin of the University of Colorado, who was not involved in the studies, said in a commentary published in the New England Journal of Medicine.
The journal is carrying two reports about two experimental cystic fibrosis drugs, both developed by Vertex Pharmaceuticals. The findings are also being presented at the North American Cystic Fibrosis Conference in Denver.
“Within 24 hours of taking the pill combination, patients were puffing less. Their sinuses were clearer. They had more energy,” Dr. Jennifer Taylor-Cousar of National Jewish Health in Denver, who led one of the study teams, told NBC News.
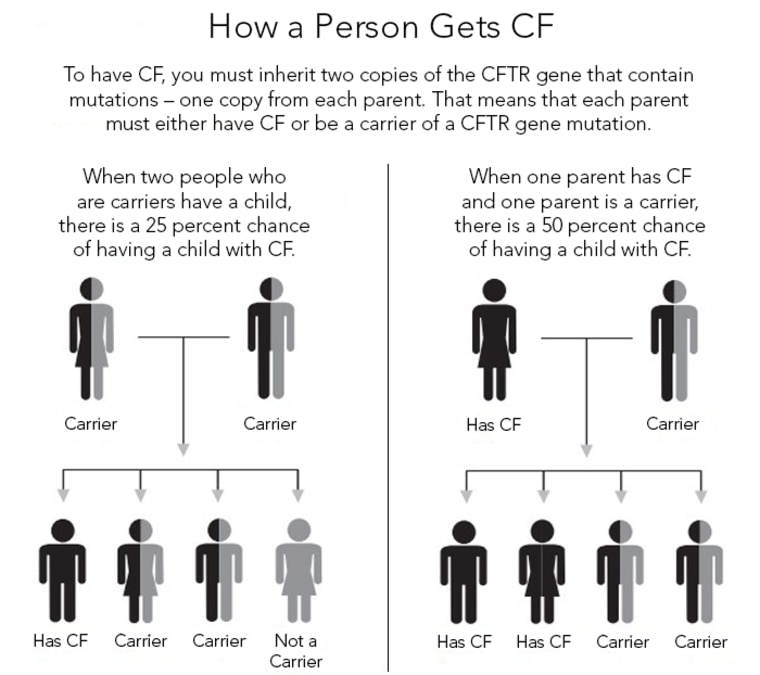
Cystic fibrosis is rare, affecting just 80,000 people globally. More than 1,000 mutations in the CFTR gene can cause the condition by affecting specific functions of cells in the lungs, digestive tract and elsewhere. People of Northern European descent are the most likely to carry these mutations, and if two carriers have a child, that child has a 25 percent chance of developing the disease.
Most commonly, patients suffer from frequent lung infections and a buildup of mucus that makes it hard to breathe. Patients usually die before they reach middle age.
New drugs can help correct some of the malfunctions in the cell, but how well they work depends on the particular genetic mutation causing a patient’s disease.
One two-drug combination won FDA approval earlier this year for patients with specific mutations. Patient groups complained when it was priced at $292,000 a year.
Researchers tested two experimental Vertex drugs among patients taking another dual combination of the drugs lumacaftor and tezacaftor. In both trials, patients around the world got either the combination with one of the two new drugs, or a placebo.
The value of the third drug is that it helps correct a genetic malfunction by adding a synergistic effect to the dual combinations, the researchers said.
“This approach has the potential to treat the underlying cause of cystic fibrosis in approximately 90 percent of patients,” Taylor-Cousar and colleagues wrote.
In the two trials of the two experimental Vertex drugs, lung function improved by between 11 percent and 13 percent, the two teams reported.
This might not seem like much, but it adds up, cystic fibrosis experts say.
“From every cystic fibrosis trial we have learned, if you could improve lung function even marginally the other clinical outcomes tracked with that,” said Dr. Christopher Richards, a cystic fibrosis specialist at Massachusetts General Hospital who was not involved in the trials.
“These are going to be huge improvements for patients. What we don’t know from these studies are the clinical outcomes — exacerbations, hospitalizations. But from other trials we know if you are able to improve lung function on the scale that they’ve done here, that should correlate with improvement in clinical outcomes.”
“We anticipate that people who will start this drug earlier, as children, are going to live normal lives with the exception of having to take these pills every day."
In 2012 Vertex won FDA approval for Kalydeco, known generically as ivacaftor. It was the first drug to address the underlying cause of cystic fibrosis symptoms. The company has been working to develop drugs that address other mutations, and has been testing them in various patients groups to get approval to use the drugs to treat more people.
“Kalydeco alone improved lung function by 10 percent,” Taylor-Cousar told NBC News.
“It was a game changer. It reduced time to need antibiotics, hospitalizations, and substantially reduced the rate of exacerbation by 50 to 60 percent.”
Adding the new drugs should improve these effects, she said. “Ultimately, we’re hoping to see longer survival,” she said.
And earlier treatment may be better. “It's the lung damage that causes cystic fibrosis patients to die young, so if we can prevent damage early on, we anticipate those patients could live a normal lifespan,” Taylor-Cousar said.
The findings are still early stage. The drugs must be tested in larger groups of people, over longer times, to determine if they have continued benefits. And, the researchers caution, it would be years before the long-term effects would be known.
But Vertex hopes to seek FDA approval for at least one of the drugs by next year.
“We anticipate that people who will start this drug earlier, as children, are going to live normal lives with the exception of having to take these pills every day,” Taylor-Cousar said.


