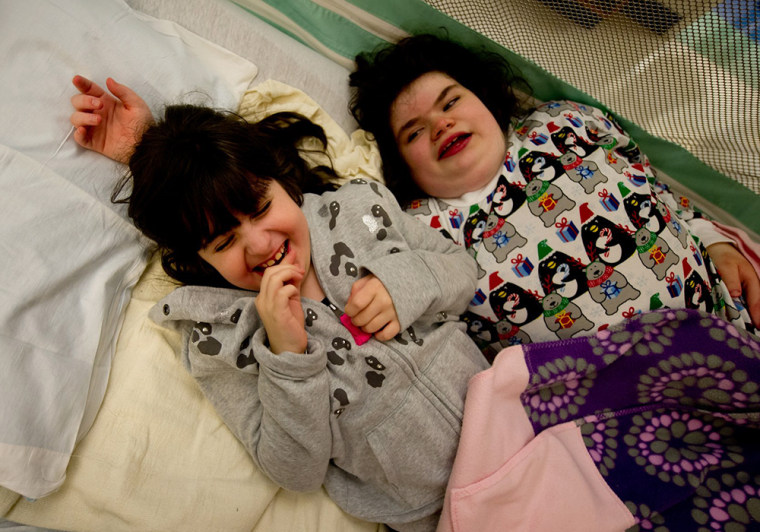
At 15, Hayley Mogul lacks the fine motor skills needed to write. Her sister Bari is 9 and still eating baby food.
There's no cure for their rare disorders, caused by unique genetic mutations. But for once, there's an advantage to having conditions so rare that drug companies cannot even think of looking for a cure. The sisters are taking part in a whole new kind of experiment in which scientists are literally turning back the clock on their cells.
They’re using an experimental technique to transform the cells into embryonic form, and then growing these baby cells in lab dishes.
The goal is the get the cells to misfire in the lab in just the same way they are in Hayley’s and Bari’s bodies. It’s a new marriage of genetics and stem cell research, and represents one of the most promising applications of so-called pluripotent stem cells.
“One day these two girls will probably change the face of medicine as we know it,” said their father, Steven Mogul.
Steven and Robyn Mogul don’t understand why both their daughters ended up with the rare mutations, which cause a range of neurological and metabolic problems.
“We have been tested,” said Mogul, a 45-year-old wealth manager living in Chicago. “We don’t have any mutations, and there are no developmental issues. We have no idea how it happened. “
The girls need special schooling and physical therapy. They must wear diapers, and when they get a cold or the flu, they can develop dangerously low blood sugar. “When the kids get sick, get colds or flu, we have to get them to the hospital,” Mogul said.
Hayley, 15, has a mutation in a gene called RAI1, which can cause Smith-Magenis syndrome. The syndrome affects 1 in 25,000 people and can disturb sleep patterns, cause obesity and behavioral issues. But Hayley’s mutation is unique and puzzling. Bari, 9, has an RAI1 mutation and a similarly unique mutation in the GRIN2B gene, which can cause learning disabilities.
“Bari doesn’t talk,” Mogul said. “She walks around, she gets around and lets you know what she wants. She is eating baby food and she is drinking from bottles.”
Hayley can attend school and can read, but lacks the fine motor skills needed to write. It’s especially unusual for two children in the same family to end up with such rare, and different, mutations.
“You ask about it from time to time. Then you know what? You just go on day by day and you just do what’s right,” says Mogul.

For the Mogul family, what’s right has been enrolling the girls in scientific studies. They’ve just been accepted in a new collaboration between the National Institutes of Health’s Undiagnosed Disease Program and the New York Stem Cell Foundation Research Institute.
Scientists at the stem cell institute will generate batches of stem cell from 100 patients on the NIH list using a new automated system, then study them to better understand their conditions.
It may sound a bit impersonal, but it’s precisely what the once-controversial field of embryonic stem cell research has been aiming to do: take an individual’s cells, grow them in the lab, study them, and test therapies on them.
Embryonic stem cells are the body’s master cells. They make up the ball of cells called a blastocyst that develops just days after conception, and each one of the cells has the power to develop into every kind of cell and tissue in the body – blood, bone, brain or muscle.
In recent years, scientists have learned how to take ordinary cells from a grown person and reprogram them to look and act precisely like embryonic stem cells. They’re called induced pluripotent stem cells, or iPS cells.
In Hayley and Bari’s case, researchers will take their cells, which carry their unique genetic mutations, and play with them in the lab. In fact, another team is already doing that.
Vasil Galat of Children's Hospital of Chicago and Northwestern University has converted skin cells from Hayley and Bari into nerve cells.
“To study these mutations we need to study how specifically they impact development of the neural system,” Galat told NBC News. “You need neural cells. You can’t take them from the patient directly. You can take skin cells or blood cells.”
He wants to see just how the genetic mutations make the nerve cells misfire, and test different drugs on them to see if it’s possible to make the cells behave more normally.
But making and growing iPS cells is very tricky, as much an art as a science. Not many people can do it. Susan Solomon of the New York Stem Cell Foundation says her organization had found a way to automate the process. They hope to grow many batches of cells. “You have to be able to look at things in parallel on a large scale,” she says.

“You can take something that looks impractical and peel it back layer by layer until you can recover what the mechanisms are.”
Galat knows at a broad level what’s wrong. But each change in the DNA can change a cell’s function dramatically, and it’s only by studying the tissue in the lab that he and the experts in New York will be able to get to the bottom of the genetic mystery.
Because Hayley and Bari’s conditions are so very rare, it doesn’t make financial sense for a drug company to do the work. And it’s so experimental that only a few labs are trying it.
Mogul doesn’t kid himself. While hopes for a treatment or cure, he knows the research isn’t going to lead to a quick breakthrough. And he doesn’t mind if one of the many scientists studying his daughters’ cells patents some discovery in the future.
“My wife and I, Robyn and I, we are not concerned about the ownership,” Mogul said. “We are more concerned about doing something positive for humankind.”