IE 11 is not supported. For an optimal experience visit our site on another browser.
UP NEXT
Prosecution, defense paint competing visions of Trump as trial begins 03:26
Surge of Chinese migrants crossing Southern border into the U.S. 02:42
Dozens of pro-Palestinian protesters arrested at Yale 02:52
Volkswagen workers vote to unionize in Tennessee assembly plant 01:32
Barry Manilow makes history with Radio City performance 01:45
Trump defense mirrors his campaign style 00:45
Senate poised to vote on new aid for Israel, Ukraine and Taiwan 00:40
TikTok faces potential ban in bill expected to pass in Senate 01:45
Suspected intoxicated driver kills two children after crashing into birthday party 02:23
40 years after ‘Footloose,’ Kevin Bacon returns to the film’s high school ahead of its demolition 02:40
Higher airfares expected as U.S. airlines project delivery of fewer planes 01:53
Good Samaritans save man from fiery crash on side of highway moments before fire reaches him 01:22
Speaker Mike Johnson’s job threatened by hardline House conservatives 01:36
Columbia University rabbi warns Jewish students, ‘return home as soon as possible’ 02:02
Opening statements set to begin in Trump criminal trial 02:16
25 years later, Columbine’s effects on school security endure with lasting impacts on students 02:28
College coach surprises basketball player by flying in his family to see him play for the first time 03:19
Officials: No criminal charges in caught-on-camera case of black bear cubs pulled from tree 02:09
Suspected counterfeit botox now linked to adverse reactions in 11 states 01:38
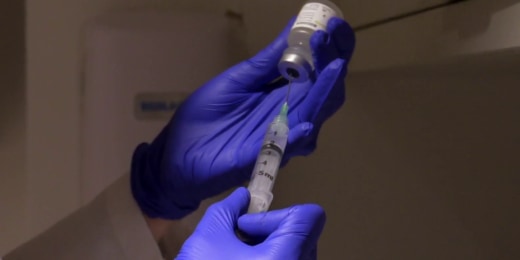
Experimental coronavirus drug remdesivir gets emergency FDA authorization 02:15 Gilead Sciences, the company behind the antiviral drug, says it will be donating 1.5 million vials to the federal government. Early results from the NIH controlled trial show patients taking remdesivir improved faster than those taking a placebo, but the full study has not been published. May 1, 2020
Read More UP NEXT
Prosecution, defense paint competing visions of Trump as trial begins 03:26
Surge of Chinese migrants crossing Southern border into the U.S. 02:42
Dozens of pro-Palestinian protesters arrested at Yale 02:52
Volkswagen workers vote to unionize in Tennessee assembly plant 01:32
Barry Manilow makes history with Radio City performance 01:45
Trump defense mirrors his campaign style 00:45