It was late last summer when Dr. Guillermo Amescua started noticing "something weird" about the eye infections he was seeing in his clinic.
Amescua, a cornea specialist at the Bascom Palmer Eye Institute of the University of Miami Miller School of Medicine, had been well-versed in using antibiotics to treat bacterial eye infections. The warm Miami weather, he said, often coaxes people into relaxing at the pool or beach before removing their contact lenses, giving bacteria — Pseudomonas, mostly — a perfect breeding ground.
Once infected, patients say their eyes are painful. They can't see clearly. They're sensitive to light. It's not often an issue, Amescua said. "We usually take care of it," he said. "There's no problem."
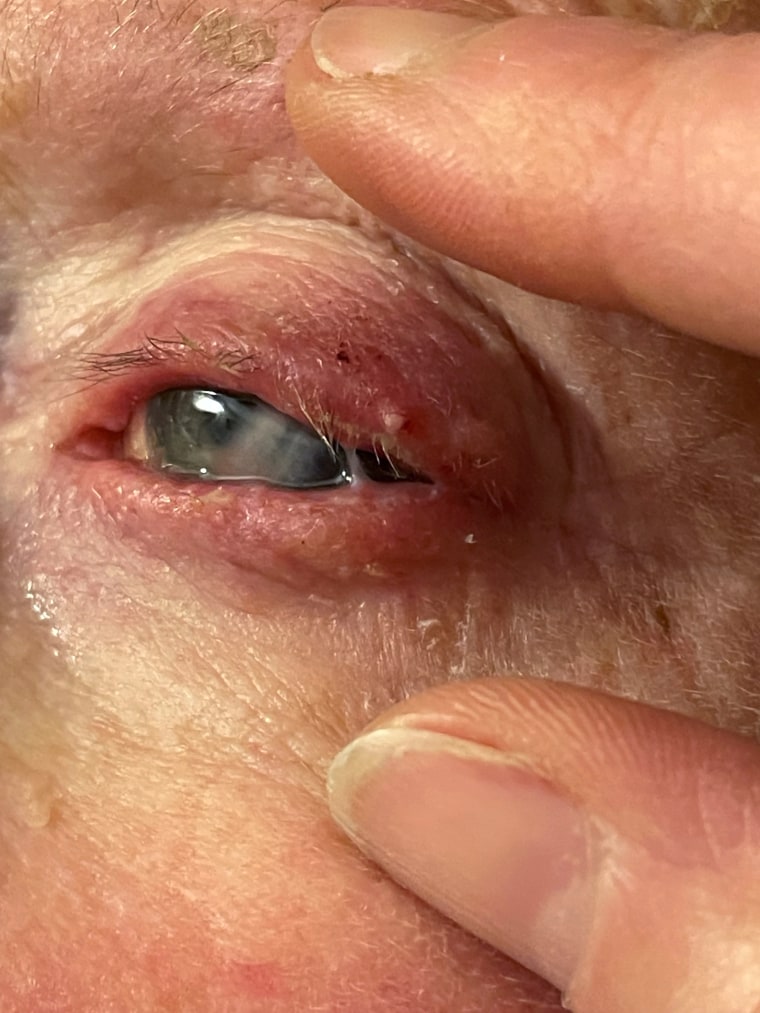
But his arsenal of antibiotics have stopped working. Over the past six months, Amescua said he has encountered at least seven cases of Pseudomonas bacteria resistant to antibiotics, resulting in devastating outcomes. The new strain is a type Pseudomonas aeruginosa that has never been seen before in the U.S., according to the Centers for Disease Control and Prevention.
Patients have needed corneal transplants. Some were blinded.
"I was getting complication after complication," Amescua said. "I don't remember ever in my 10-plus years losing the battle against a Pseudomonas infection so many times in such a short period of time."
Among cases nationwide, the CDC says the common thread appears to be a particular brand of eyedrops: EzriCare Artificial Tears. The agency urged people to stop using the eyedrops immediately.

Most were purchased online, but at least one person reported buying the drops at Costco, the CDC told NBC News this week.
As of Wednesday, the artificial tears, manufactured in India by Global Pharma Healthcare, have been linked to at least 55 cases of antibiotic-resistant Pseudomonas infections nationwide. Cases so far have been reported in 12 states: California, Colorado, Connecticut, Florida, New Jersey, New Mexico, New York, Nevada, Texas, Utah, Washington and Wisconsin. The company has since recalled its products, also sold under the brand name Delsam Pharma.
One person has died and five other people have been blinded, at least in one eye, the CDC said. The agency first reported potential problems with the eyedrops on Jan. 20, but did not issue a public health alert on the matter until Feb. 1.
"This was a very complex investigation that required a number of techniques that are time consuming and challenging to coordinate and implement," a CDC spokesperson told NBC News. "Due to the widespread nature of this outbreak, the investigation required coordination and data collection from a large number of state and local health jurisdictions."
One suspected case involves 81-year-old Judy Gregory, of Elsmere, Kentucky. Gregory said she used EzriCare Artificial Tears to ease her dry eyes before she was hospitalized with an eye infection June 1, 2022. Her daughter, Kim Harrison, said her mother's infection was so bad, her doctors "said it would be a miracle if they could save the eye."
Fortunately, they did. But eight months later, Gregory says she continues to suffer from vision loss. Harrison still takes her mother to eye appointments because Gregory's impaired vision means she can no longer drive. Gregory said her doctor has reported the case to health officials.
While the CDC has not yet definitively traced the infections to the eyedrops, the agency is investigating cases with the Food and Drug Administration and state and local health officials.
The CDC is now testing unopened bottles of EzriCare Artificial Tears to determine "whether contamination may have occurred during manufacturing," the agency said.
The FDA first learned of the Pseudomonas aeruginosa outbreak in December 2022, a spokesperson told NBC News. At that time, many potential products were being investigated by the CDC. The FDA learned in January that the Pseudomonas aeruginosa outbreak was associated with EzriCare Artificial Tears. Over-the-counter eyedrops do not require FDA approval.
Darlene Miller, who heads the Bascom Palmer Eye Institute lab, urged people who may have been infected by the drops to contact a physician immediately.
The bacteria, Miller said, "can destroy the eye within 48 hours."
According to the CDC, symptoms of an eye infection include:
- Yellow, green or clear discharge from the eye.
- Eye pain or discomfort.
- Redness of the eye or eyelid.
- Feeling of something in your eye (foreign body sensation).
- Increased sensitivity to light.
- Blurry vision.
Meanwhile, doctors like Amescua worry that people are still using the contaminated eyedrops.
"It's alarming because it has resistance to pretty much everything we have," he said.
Follow NBC HEALTH on Twitter & Facebook.
