Geneticists say they've built a working yeast chromosome from the bottom up for the first time — a feat that could open the way for custom-made biofactories that churn out fuels and pharmaceuticals.
"We think this is going to be critical as we transition as a species from a petroleum economy to a bio-economy," said Jef Boeke, a researcher at NYU Langone Medical Center who pioneered the project.
Boeke and his colleagues laid out the procedure in a paper published online Thursday by the journal Science.
The work follows up on past achievements in synthetic biology, including the creation of the first synthetic bacterial cells in 2010. The international project in which Boeke is involved, known as Sc2.0, is different in that it's reconstructing an organism's genetic code piece by piece, gradually remaking the machinery to control a cell rather than plugging in an entire genome at once.
The fact that the cell happens to be brewer's yeast, known scientifically as Saccharomyces cerevisiae, opens many more doors to potential applications. Genetically tweaked versions of yeast are already being used to make biofuel as well as ingredients for an anti-malaria drug.
Boeke said the technique that was used to create a chromosome could provide more opportunities for that kind of bio-manufacturing. He's even looking into creating entirely new yeast chromosomes that incorporate human genetic code for medical screening purposes.
"This is significant as an example of synthetic genomes aimed well beyond making mere copies of chromosomes," Harvard geneticist George Church, who was not involved in the project, told NBC News. He said the study reflected a new trend toward "making significant functional changes — ideally, changes useful for biotech productivity and safety."
How it was done
Chromosomes are strings of genes that contain the instructions for all the functions within a cell. Human cells typically contain 23 pairs of chromosomes. Yeast cells have 16 pairs. The Science paper describes the reconstruction of yeast chromosome III, which regulates mating behavior and other functions.
The project took seven years to complete, starting with the assembly of tiny bits of genetic code by more than 100 students who participated in a program called "Build-a-Genome."
Boeke compared the job to writing a crowdsourced book, where every letter of the book is a DNA base pair. At the lowest level, 50 to 75 base-pair letters were combined into words. Those words were strung together into paragraphs consisting of 750 to 1,000 base pairs. The paragraphs were combined into 3,000-word pages, and then the pages were assembled into 11 separate chapters of genetic code.
Each chapter was edited to change or delete code. The researchers omitted what appeared to be needless words — what's popularly known as "junk DNA." They also inserted strings of DNA to serve as identifying markers, as well as codes for shuffling the genes in the chromosome like sections in a loose-leaf binder.
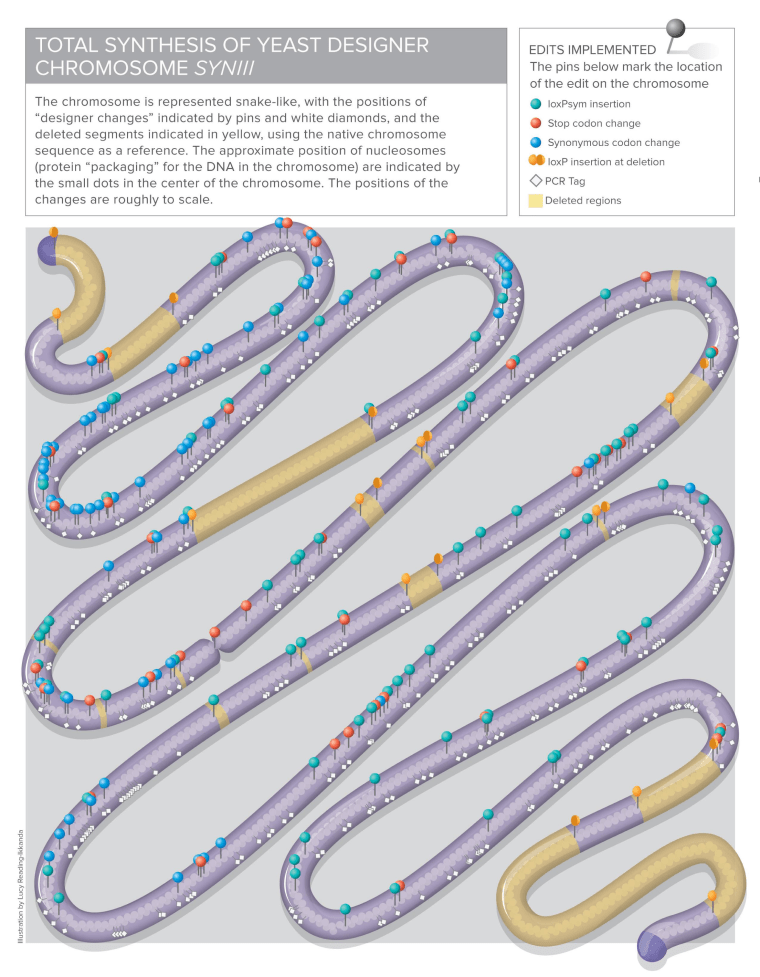
The researchers started out with 316,617 base pairs in the natural chromosome, and slimmed the code down to 272,871 base pairs. More than 500 genetic alterations were made along the way.
"We didn't know ahead of time whether it was going to work out or not," Boeke said. "We know of many instances where we make one single base change and it will kill the yeast dead."
Boeke and his colleagues swapped their synthetic code into live yeast cells, chapter by chapter, to check their work. They were relieved to find that the yeast cells with the synthetic chromosome, called SynIII, generally functioned as well as cells with the natural chromosome.
Then the researchers used a technology called SCRaMbLE (Synthetic Chromosome Recombination and Modification by LoxP-mediated Evolution) to shuffle the genes in the synthetic chromosome. They "scrambled" the genes to turn a sterile yeast cell into a yeast cell that could mate.
"It shows that, using the scrambling system, we can really transfer a new property to the yeast," Boeke told NBC News. "I think we're going to be able to show many more examples of this in the future."
Perils and possibilities
Boeke said SynIII marked only the beginning of a total yeast makeover. "We still have 15 chromosomes to go," he noted. The entire task could take at least a couple of years to complete.
In an email exchange with NBC News, Church noted that the techniques described in the Science paper were complementary to other strategies for accelerated evolution, such as MAGE and CRISPR. "The impacts (good and bad) typically do not become significant until these strategies are applied genome-wide," Church said.
Boeke noted that the yeast strains used for the Sc2.0 project have been specifically designed for laboratory use, and have to be fed nutrients to survive. "In nature, such strains don't do well in the world at all," he said. That's just one of the safeguards aimed at keeping undesirable mutant yeast strains from getting into the wild.
"We're very careful to build safety into the projects," Boeke said.
In the future, the yeast genome could be tweaked to make the cells more tolerant to alcohol — opening the way to more efficient biofuel production. Boeke said he's also studying ways to create neo-chromosomes, or "add-on" chromosomes, that incorporate custom-made genetic code for a wide variety of applications.
"One that we're pretty excited about is a network of human genes that we can bring into the yeast," Boeke said. "These genes are involved in a series of unsolved metabolic diseases that there's no treatment for. We think that by bringing this neo-chromosome into the yeast, we might be able to develop some good models to understand those diseases better, and perhaps even to develop some therapeutic agents. I do work at a medical school, so I'm always paying attention to these applications."
Update for 3:45 p.m. ET March 27: In an emailed statement, genomics pioneer Craig Venter and his colleagues at the J. Craig Venter Institute said they applauded the newly published work. Venter's team was behind the 2010 creation of the synthetic bacterial cell. Here are a couple of excerpts from the statement, attributed to Venter as well as Hamilton Smith, Clyde Hutchison, Dan Gibson and John Glass:
"Advances such as these are important enabling steps for the entire field of synthetic biology. This work is another remarkable example of how synthetic biology can be used to rewrite chromosome sequences at a sizable scale. ..."Each development in our field not only enhances our general understanding of biology, but also creates innovative steps upon which all of us can build and expand. We continue to believe that synthetic biology advances will form the basis for a new world economy and will positively impact all aspects of society."
Boeke and Srinivasan Chandrasegaran of Johns Hopkins University are among 80 authors of the paper published online by Science, titled "Total Synthesis of a Functional Designer Eukaryotic Chromosome."
