Nine-year-old Killian Owen was the first child to try an experimental leukemia treatment that was showing promise in adults — but the chance came too late. Yet the youngster left a precious legacy: Scientists are using his preserved cells to help create stronger drugs for other children desperate for new options.
Killian’s saga illustrates both the pitfalls and promise of pediatric cancer research. Scientists are finding exciting leads, but few novel therapies for children are in the pipeline. When promising ones do come along, sick youngsters often must wait years for safety testing in adults to be done before they get a shot.
'One step behind'
“We’re always one step behind,” sighs Dr. Alan Wayne of the National Cancer Institute, who treated Killian with a drug called BL22 and is pushing for quicker clinical trials for children.
“The fact he was given a chance has led to so much hope for so many other children,” says Killian’s mother, Grainne Owen, who began a charity to spur pediatric cancer research. But, she adds, “You shouldn’t have to jump through the hoops we had to jump through to get our child to try a new drug.”
There are signs that may be changing.
In a first, the Food and Drug Administration recently gave fast-track approval for a cancer drug for children to begin selling even before testing in adults is finished. The drug, Clolar, is to treat relapsed leukemia. FDA’s drug chief, Dr. Janet Woodcock, says she’s open to speeding through more child-first cancer treatments whenever those smallest patients have no good options.
And a stark new report from the influential Institute of Medicine urges government and drug makers to cut children’s waiting time for access to experimental therapies — and for the NCI to actually develop those drugs if industry sees too little profit to do so.
A “near absence” of pediatric cancer drug research “threatens to halt the progress in childhood cancer treatment achieved during the past four decades,” the report warns.
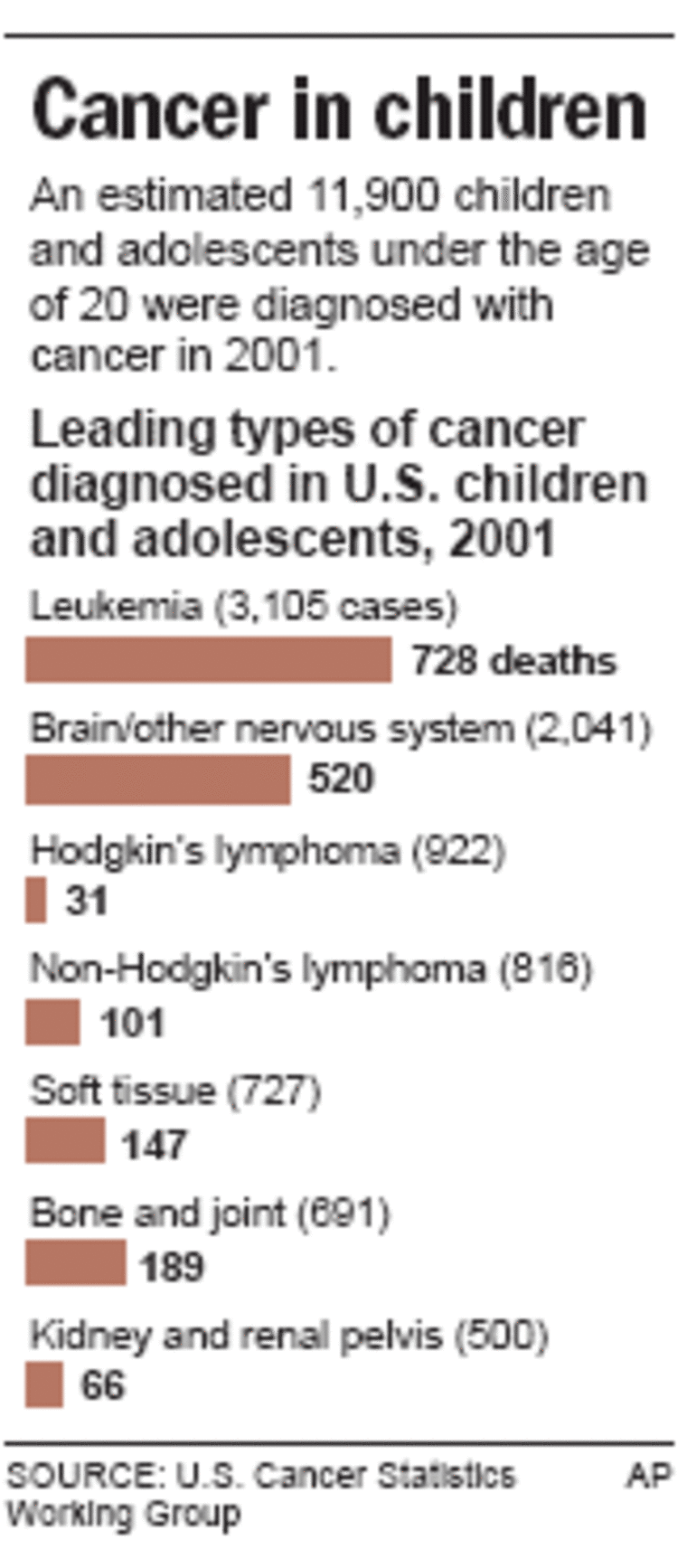
Childhood cancer survival is at an all-time high, with 80 percent of patients now cured. But more than 2,000 children a year still die, making cancer the leading pediatric killer disease.
While much of the progress came from mixing-and-matching adult chemotherapies in young patients, specialists agree it will take more novel approaches to help the remaining hardest-to-treat children. That’s because even cancers with the same name can act differently in children than in adults.
Little incentive for drug companies
But there are so many more adult patients that it’s not profitable for drug companies to pursue pediatric cancer aggressively, the IOM report says. Indeed, a recent industry survey shows 32 products being tested for childhood cancers; about half are already sold for adults, and the rest are in more advanced testing for adults than for children.
“If a drug cures 300 cancers a year, who’s going to pay for that drug?” asks Dr. Lee Helman, NCI’s pediatric oncology chief. “It’s going to be a societal problem.”
Still, scientists hot on the trail of so-called targeted therapy — ways to attack the molecular differences that make some cancers more aggressive — are trying to speed children’s access to early stage research studies. Some examples:
- Johns Hopkins University is about to start pediatric testing of a drug that blocks a mutation in a gene called FLT-3 that slashes survival for patients with acute myeloid leukemia, or AML. Dr. Donald Small urged drug maker Cephalon Inc. to allow the child study because soon-to-finish adult tests were showing promise.
- The NCI just began administering the highest child dose possible of BL22, in the study Killian Owen pioneered, based on analyses of the Atlanta boy’s preserved cells that found he had been given too small a dose to work. Later this year, NCI plans the first tests of a more potent version, HA22, in adults and children simultaneously. The experimental antibodies bind to a protein on cells of some patients with acute lymphoblastic leukemia, or ALL, and deliver a cancer-killing toxin.
Killian’s saga shows why awaiting adult research can backfire: He was given a fraction of the adult BL22 dose, standard procedure for early pediatric testing even though children’s faster metabolism often requires a higher dose, explains Wayne.
“Clearly the Owens made progress in opening the logjam,” for children, he says. “The pendulum is swinging. ... The problem is it’s never fast enough.”
