Deep brain stimulation can offer relief to patients with diseases such as Parkinson’s and epilepsy, but it requires brain surgery to implant electrodes deep inside the head.
Now researchers think they’ve come up with a way to do the same thing without cutting. They have used electrodes on the heads of mice to make the animals wiggle their paws and whiskers — even under deep sedation.
Researcher think it’s safe enough to eventually try out on people.
It works because the brain is a natural conductor of electricity, said Ed Boyden, co-director of the M.I.T. Center for Neurobiological Engineering.
“All of our knowledge about the brain says it an electrical conductor,” Boyden said.
Boyden and colleagues call the method temporal interference, and it allows them to send electrical signals deep into the brain without affecting the layers in between.
Related: Brain Stimulation Technique Might Treat Addiction
It works on the same basis as other types of wave interference, from the noise-cancelling headphones that let you sleep on a noisy airplane to light-cancelling experiments that delight schoolchildren.
It has always been possible to send electrical signals into the brain. The trouble is high-frequency signals just go right through it.
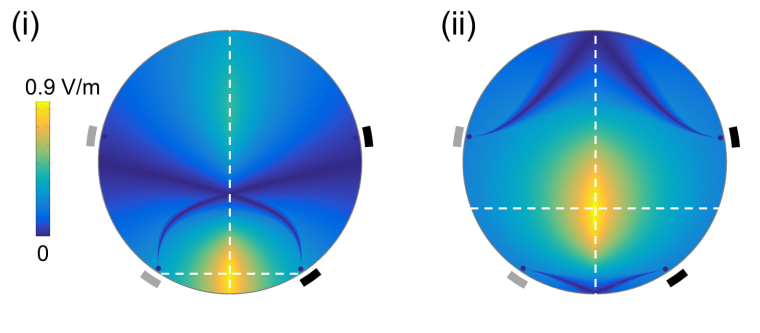
“When you put these two high-frequency signals at the top of the brain, you get a different frequency in the middle of the brain."
Low-frequency signals can affect brain cells but if delivered from the outside, they first hit the outer layer, known as the cortex. To treat conditions such as Parkinson’s, you have to get into the deeper layers.
What Boyden’s team did is use the principle of interference to turn two high-frequency signals into one, targeted, low-frequency signal.
Related: Brain Pacemaker Benefits Parkinson's
“When you put these two high-frequency signals at the top of the brain, you get a different frequency in the middle of the brain,” Boyden said.
So sending a 2,000 kilohertz signal from one direction, and a 2,001 kHz signal from another will result in a 1 Hz signal, Boyden said.
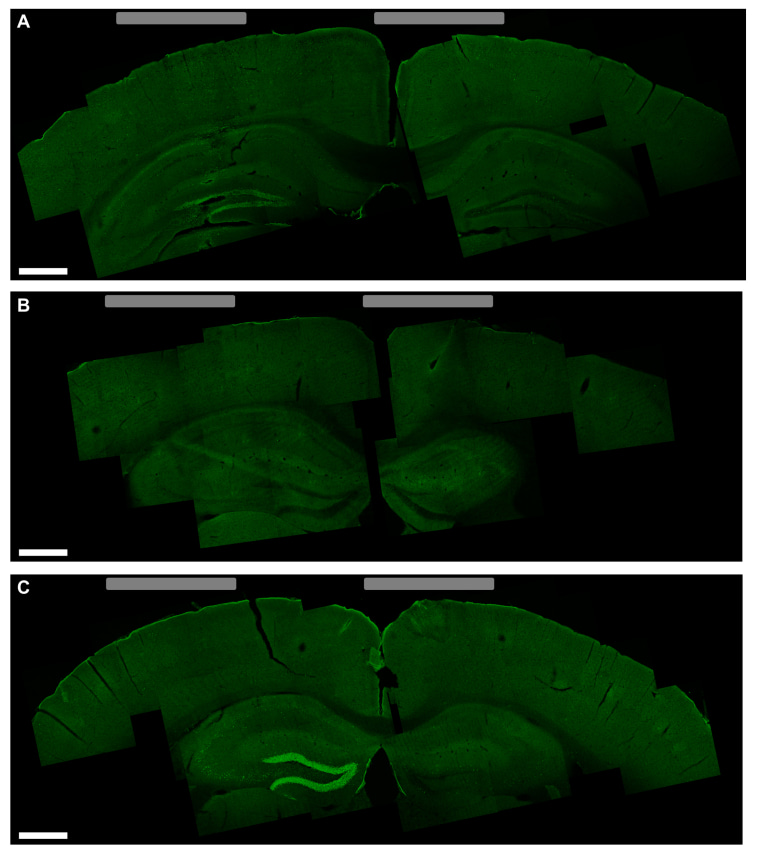
They tried this is mice, which were anesthetized. Writing in the journal Cell, the team said they could excite neuron activity with their signals.
Then they aimed at motor cortex areas that are well-studied in lab mice and got the unconscious mice to wiggle their paws and whiskers.
Related: Scientists Control Mice With a Switch
It’s a long way off from being tried on people.
“We have to make it more precise,” Boyden said.
“Future studies, perhaps using larger numbers of electrodes and multiple sets of interfering fields, may be able to pinpoint even smaller regions of the brain, or multiple regions of the brain. An open question is how small a focal volume may be achieved,” the team wrote.
And to treat Parkinson’s disease requires a range of frequencies different from those the MIT team used.
But they think it’s promising.
“Given the remarkable therapeutic benefits of deep brain stimulation for patients with otherwise treatment-resistant movement and affective (mood) disorders the prospects for noninvasive deep brain stimulation using electricity are potentially exciting,” they wrote.
Boyden said there’s nothing surprising about the technology. “Most technologies that work fairly well look really simple and obvious in hindsight,” he said.
